FDA Clears Apple Studio Display XDR for Medical Imaging
Toggle Dark Mode
Apple’s new Studio Display XDR just received a huge vote of confidence from the US Government, as the Food and Drug Administration (FDA) announced it’s now cleared to be used for diagnostic medical imaging.
While that’s a big deal for the medical community, it also proves that the new XDR display isn’t just ideal for professional photo and video editing workflows, but has the level of color and light accuracy and reliability needed to handle people’s lives.
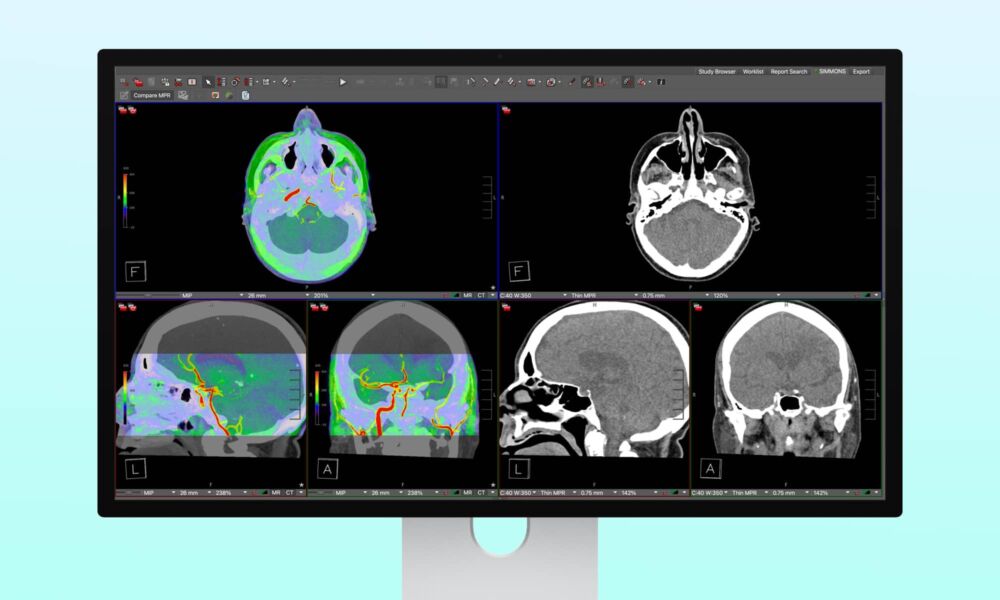
The new feature serves as one of the 16 Reference Modes available on the Apple Studio Display XDR. When Apple unveiled the new display in early March, it listed “Medical Imaging (DICOM-350 nits)” and “Medical Imaging (DICOM-600 nits)” among those modes, but added a footnote that they were only “intended for use by medical professionals,” and “not intended for use in mammography.”
More significantly, they also required the use of a “Medical Imaging Calibrator” tool in macOS that wasn’t yet available. Apple described the medical imaging capabilities in more detail in its Studio Display XDR Technology Overview, while also tagging it with “Expected by April 2026.”
In a first for Apple displays, Studio Display XDR supports medical imaging for diagnostic radiology when using the new medical imaging presets and Medical Imaging Calibrator within macOS. The feature is intended for trained medical professionals for use in general radiology, excluding mammography. To meet the needs and preferences of radiologists, two new medical imaging reference modes are built into the display, with unique capabilities.
As a medical diagnostic tool, this naturally requires FDA clearance. However, it seems it’s not the Apple Studio Display XDR itself that’s the issue, but rather the “Medical Imaging Calibration Feature (MICF)” that the FDA cares about. The agency gave it a green light on April 1. That’s about four months after its original submission on November 17, 2025, giving us an idea of how far ahead Apple has to plan for these types of features.
Greg Joswiak, Apple’s senior VP of worldwide product marketing, also shared the news, confirming that the MIC tool will be available as part of macOS 26.4.
The Medical Imaging Calibration feature for Studio Display XDR is now FDA cleared & available on macOS 26.4 in the US! That means radiologists can use the world’s best pro display in general radiology. Incredible to see the intersection of health & technology. ? pic.twitter.com/vXDIFLnHeW— Greg Joswiak (@gregjoz) April 7, 2026
That’s not a huge jump, considering the Apple Studio Display XDR officially requires at least macOS 26.3.1. While it’s ultimately just a Thunderbolt Display under the hood, folks running older versions of macOS won’t be able to take full advantage of its capabilities, whether that’s the high-quality Reference Modes for pro creatives, or even the Center Stage camera and Spatial Audio speakers.
While the Studio Display XDR starts at $2,899 for a version with the VESA mount adapter, or $3,299 if you want a stand, that only seems pricey for those of us who don’t work in professional creative or medical fields. A high-end 12MP diagnostic display can easily run over $30,000. That’s technically a lower resolution than what’s offered by Apple’s 5K display, although it’s fair to say these distinguish themselves in other ways, and can handle tasks which Apple’s display still cannot, such as mammography.
It’s also worth adding that the FDA clearance naturally only applies to the use of the Studio Display XDR for radiologists in the United States. While Apple could pursue similar clearances from other health authorities around the world, it’s telling that the Apple Studio Display XDR specs in Canada and other countries like the UK, Australia, Japan, and Germany omit any mention of the Medical Imaging reference modes.







